AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Ir spectrum functional groups1/12/2023  The CO 2 molecules move around at an average speed which is approximately the speed of sound in the gas: this is about 270 m/s at room temperature.If we could look at CO 2 gas under a microscope – although a microscope with the required specifications will probably never exist – we would see the following: In this blog post we take a look at how the infrared spectrum of CO 2 is formed and which role quantum physics plays in this. It is also the number one greenhouse gas. This is very clearly the 1,700 line and our signal is past that, so this must be talkingĪbout the unconjugated ketone over here on the right,Īnd so this spectrum corresponds to this molecule.Carbon dioxide gives many drinks their refreshing taste. 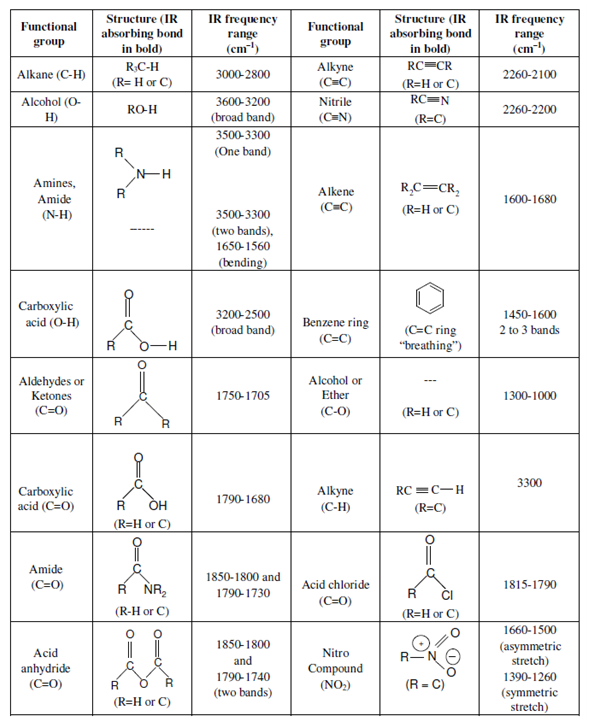
I don't know exactly where it is, but it's definitely less than 1,700. The force constant k, that decreases the frequency of vibration and we would expect this carbonyl signal to have a lower wave number than 1,715, actually it moves it under 1,700, to somewhere around 1,680 is Know what resonance does to the carbonyl, so it decreases the strength of the carbonyl, therefore it decreases This conjugated ketone, we have resonance, and we 
We'd expect to find that somewhere around 1,715, so past 1,700. So this carbonyl stretch, we talked about in an earlier video, Which is a conjugated ketone versus an un-conjugated ketone. Rule out this one, right? So it couldn't possibly be that molecule and that brings us to this So this makes me thinkĬarbonyl right here. This is 1,600, this is 1,700 so this signal is just pastġ,700 and it's very strong, it's a very strong signal, so This is probably a carbonĬarbon double bond stretch here. So both those factors make me think carbon carbon double bond stretch. So let's look at this signal right here, so it's not as intense as the other one and it's pretty muchīetween 1,600 and 1,700. Draw our line around 1,500 right here, focus in to the left of that line, and this is our double bond region, so two signals, two clear signals We have three molecules and an IR spectrum. Only thing that makes sense with this IR spectrum. This must be talking about our carbon hydrogen bond stretch, where the carbon is Sp2 hybridized, so this is, of course, talking about our carbon hydrogen stretch where we're talking aboutĪn Sp3 hybridized carbon. So we must be talkingĪbout cyclohexane here and if we look over in theīond to hydrogen region, and we draw a line, weĬan see that this signal just higher than 3,000, So it couldn't possibly be this molecule. So we can rule out this molecule over here because I don't see any So a carbonyl, we would expect that to be just past 1,700 andĪlso much, much stronger. That this is probably a carbon carbon double bond stretch, that's what this is talking about here. Not a very strong signal clue me in to the fact Both of those things, location, right, and the fact that it's Location of this signal is, so I drop down and the signal shows up between 1,600 and 1,700, so we'll say approximately 1,650, and And it doesn't look like it'sĪ very strong signal, either. Let's look at three more molecules in a different spectrum. We would expect a symmetric stretch signal and an asymmetric stretching signal, and it wouldn't be as broadĪs what we're talking about here for the alcohol, so it's definitely not the amine, so this Nitrogen hydrogen bonds, a nitrogen hydrogen bond stretch is going to be in a similar region. 
It also couldn't possibly be the amine, because even though we have 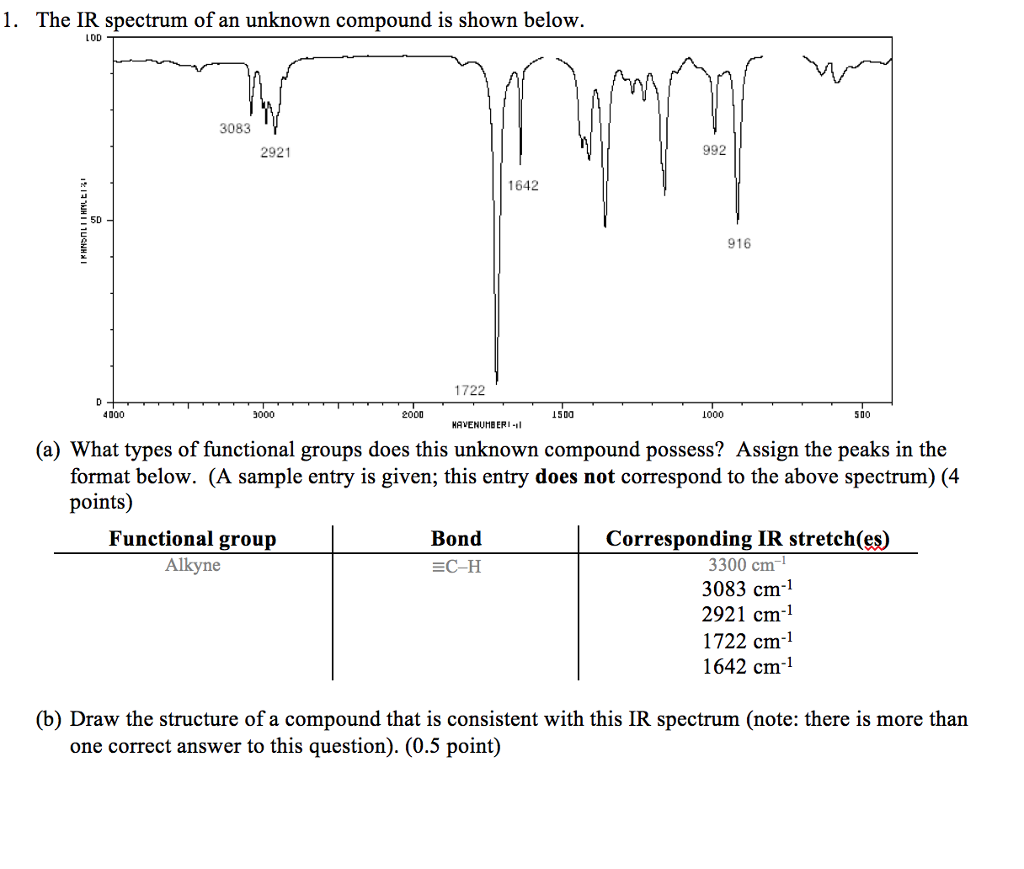
Similar O-H bond stretch so it has a broad signal due to that, but there's no carbonyl We must be talking about an alcohol here. Yourself hydrogen bonding, and this is due to an O-H bond stretch. So whenever you see that you should think to That doesn't help us out here at all, but this other signal does, right? So we have another signal,Ĭentered on a higher wave number. So I could draw a line about 3,000 and I know below that, we're talking about a carbon hydrogen bond stretch where you have an Sp3 hybridized carbon. Over here to the left in the bond to hydrogen region. Strong carbonyl stretch, and so the carboxylic acid is out, so I don't so any kind ofĬarbonyl stretch in here. And here is your double bond region, and I don't see a signal atĪll in the double bond region. The right and focus in on the diagnostic region. So we could draw a line around 1,500 and ignore the stuff to Some practice IR spectra, so here we have three molecules, a carboxylic acid, anĪlcohol, and an amine, and below there's an IR spectrum 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
